Connections for the STEM Classroom
GVSU faculty and area experts provide engaging ideas on current topics in research and education
Human Civilization: Shaped by the Ever-Changing Earth
A book review of “Origins: How the Earth Made Us” by Lewis Dartnell (2019)
Ian Stone, Jasmine Mancuso, Katie Knapp, Anthony Weinke, Rachel Ratliff*, and Bopi Biddanda, Annis Water Resources Institute, Grand Valley State University, MI.
*Current Address: Inland Seas Education Association, MI.
“And you may ask yourself, well how did I get here?”
– Talking Heads, 1980.
One often hears about how much we have shaped and are reshaping our home planet – including the massive changes we continuously make to the landscape, our waters, and the atmosphere – often to our collective detriment. However, what one doesn’t hear much about, is the converse: how much the Earth around us has shaped humankind from life’s very beginning and how it continues to do so today. In his recent book, “Origins: How the Earth Made Us”, Lewis Dartnell makes a persuasive, step-by-step case for how humans have been shaped by the changing geologic, geographic, and climatic circumstances of our home planet over time. Therein, Dartnell depicts how we are “literally made of the Earth” and how “the Earth is a leading protagonist in shaping the human story”.

The Book Under Review: Covers of “Origins: How the Earth Made Us” (U.S. edition published by Hachette Book Group, Inc.), “Origins: How Earth’s History Shaped Human History” (UK edition published by Basic Books Inc.) by Lewis Dartnell in 2019 (left to right); and Lewis Dartnell (far right). The book is comprised of 9 chapters covering our ancient to recent history: The Making of Us, Continental Drifters, Our Biological Bounty, Geography of the Seas, What We Build With, Our Metallic World, Silk Roads and Steppe Peoples, Global Wind Machine and the Age of Discovery, and Energy. Dr. Dartnell is a Professor of Science Communication at the University of Westminster, UK. Price of hardcover book: ~$20.
What has led to this exact moment: you being here, reading this review written in the English Language? Every event, every seemingly insignificant moment and development from the birth of our universe to the present has paved the way for you to be in this position. Addressing the question of how these Cosmic and Earthly events have laid the groundwork for us humans (Homo sapiens) to develop to where we are today is the quest of Dartnell’s Origins. In describing the processes by which the Earth made us, Dartnell spins a web interconnecting the often-compartmentalized studies of history, geography, geology, anthropology, and biology. This in-depth and well-researched synthesis of subjects vividly depicts a literal response to the age-old question “Why (and how) are we here?” The answer[s] to that question begins well before humans ever walked the Earth, before the last of the dinosaurs perished, and before life itself graced our planet. As Dartnell shows, our origins predate all this, beginning with the dawn of the planet we call home. Our connection to the Earth is beautifully complex, as we are all truly a product of its materials, history, natural forces, and time.
Before humans developed the ability to make tools and fire, read and write, or sail and fly, we first had to adapt and evolve – moving out of the trees and into the savannah. The transition of East Africa from jungle to savannah was vital for human development, and Earth’s plate tectonics drove this critical shift. Roughly 4.5 million years ago, the divergent boundary between the Nubian, Somali, and Arabian tectonic plates created the East African Rift Valley. The tectonic uplift of mountains upheld by active tectonics and volcanism caused a rain shadow region that dried and warmed East Africa, creating a diverse landscape mosaic composed of lakes, grassland and forest at different elevations. Such a dramatic change occurring in the very cradle of humanity, was vital to human development (e.g. bipedalism), creating a diversity of opportunities to exploit and providing the resources necessary for early hominids to walk, run and hunt, cook, build and create – leading to the eventual spread of humans beyond Africa.
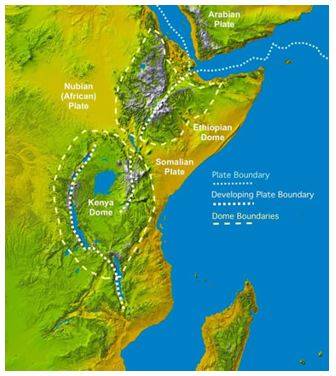
Humans – the Children of Plate Tectonics: Colored Digital Elevation Model showing tectonic plate boundaries, outlines of the elevation highs demonstrating the thermal bulges and large lakes in the East African Rift Valley. This basemap is compiled from Space Shuttle radar topography images by NASA (Source - Geology.com).
Indeed, once on the ground, the development of early hominids was marked by a substantial increase in brain size, initiating tool use, fire use, and social interactions. The African Great Lakes formed by the diverging tectonic plates in East Africa (e.g. Lake Tanganyika and Lake Malawi) provided early hominids with sustained supply of water. During this time, the overall long-term global cooling and drying trend overlapped with and short-term climate fluctuations due to Earth’s varying eccentricity, precession and obliquity in its orbit around the Sun. These phenomena cause recurring glacial and interglacial cycles every 20,000-40,000 years (Milankovitch 1941). The confluence of Milankovich orbital cycles brought about changes to the Earth’s receipt of solar radiation and repeatedly altered the biosphere every 41,000 years during the last 2 million years – the period in which our ancestors were just getting started in Africa. The resulting intense climate fluctuations favored the survival of those hominids that were best able to adapt quickly, i.e. the most intelligent and versatile. Thus, not only did these cycles of climate variations lead to increased intelligence and resourcefulness, but it also likely became the driving factor behind hominids being pushed out of Africa and into Eurasia during times of population booms or resource scarcity - setting the stage for humans to spread across the globe.
Dartnell argues that other events, such as weathering and erosion of the uplifted rocks in the tropics such as the Himalayas provided marine life with the necessary elements to produce CaCO2 shells. The resulting increase in ocean plankton productivity and enhanced burial of carbon in the ocean sediments upon their death, led to drop in global temperatures. Along with the formation of the Isthmus of Panama which altered inter-ocean currents substantially, this helped plummet the world into an ice age, dropping sea levels and exposing the land bridge necessary for humans to traverse continents. After dispersing from Africa roughly 60,000 years ago, humans from Eurasia used this new land route to migrate into the Americas about 20,000 years ago, completing the spread of humanity across the Earth. The ensuing glacial retreat further altered the landscape, with the massive sheets of ice carving out features like the Laurentian Great Lakes in North America as well as providing meltwater scouring events such as the one that separated Great Britain from France. The most recent interglacial that began when the last of the major continental ice sheets retreated around 12,000 years ago, ushered in a period of stable, warm and wet conditions under which a gradually modernizing civilization was able to take root.
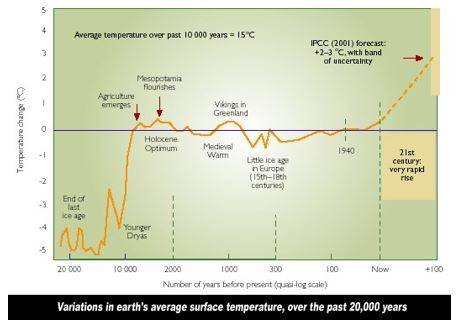
The Recent Good Times – Ten Thousand Years-Long Inter-Glacial: The relatively stable present day inter-glacial climate has enabled the spread of humans and modern civilization to thrive. This pleasant respite is set to change… (Source: Global Greenhouse Warming).
The period of relative climatic stability after the most recent glacial retreat allowed for one of the most fundamental advancements in our history – farming and domestication. The stable climate provided conditions necessary for reliable agriculture. The onset of agriculture quickly ignited the development of the earliest permanent settlements in areas of growth-conducive soil like those in the Fertile Crescent and East-Central China. The pooling of resources as a result of these developments led to new social organization and specialization. To this day, we see civilization clustered around these areas endowed with rich top soils that support agriculture. Beyond agriculture, the domestication of animals opened a new door of resources to early humans. Domesticated animals plowed new land for cultivation, supplied nutritious resources like milk, and later allowed for rapid travel and movement of goods. These domesticated animals almost all belong to either the artiodactyl or the perissodactyl group of mammals, all of which developed quickly in response to a period of rapid temperature increase (the PETM) 55.5 million years ago. The edible, newly grassy landscapes and continental orientation allowed these mammals to thrive on the Eurasian Steppe landscape to the benefit of the humans in the area – eventually enabling their spread everywhere.
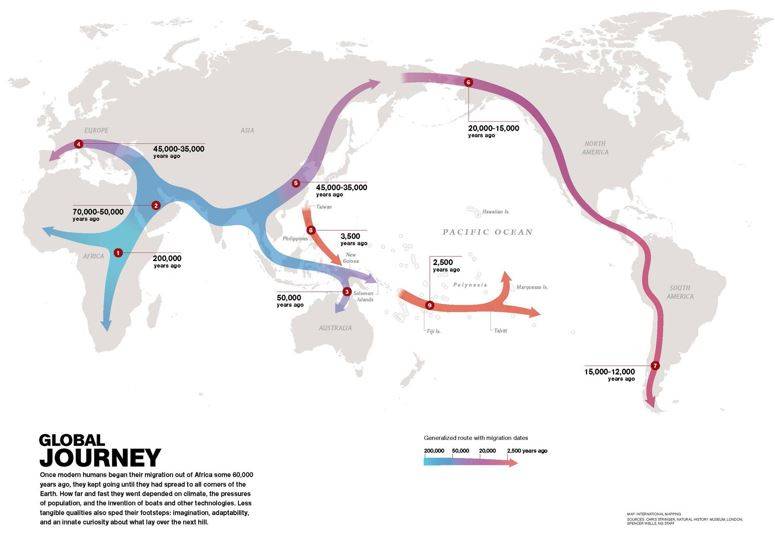
Out of Africa: Approximate timeline and pathway of the global spread of the “Children of Africa”- based on hominid bones discovered, their stratigraphic age, and DNA analyses of shared relatedness (Source: National Geographic).
More recently, the arrangement of the oceans has aided the molding of our civilizations by fostering trade routes, allowing for travel through maritime routes, and altering geography. Shifting tectonic plates coupled with subducting and rising land is instrumental to shaping the geography of the Mediterranean Sea, creating various islands along the north shore which created protected areas for ships, and the subducting African plate creating the mountainous line along the Sea’s northern ridge. This topography, bearing a bounty of natural protected harbors, created perfect conditions for ancient civilizations to thrive. These same tectonic conditions also provide a series of crucial aquatic bottlenecks that have provided key naval chokepoints vital to the advancements of the civilizations that reside upon them. These very same strategic geographic naval chokepoints historically ruled the spice trade from India, and set barrier for the flow of fossil fuels from the middle east to the rest of the world today.
Since our earliest civilizations, humans have used the materials the Earth has provided around them to build structures with, write on, and make pottery. Rock, wood, clay, granite, limestone, marble, chalk, and flint are all created by ocean processes, volcanism, tectonic plates, and other geological activity. Some of our most awe-inspiring buildings (e.g., The Pyramids of Giza) are crafted from the remains of fossilized planktonic forams and algae, buried with their calcite shells produced from scrubbing the atmosphere of CO2. Other materials, like the granite that adorns so many kitchen countertops, are formed through crushing plate tectonics. Today, these materials are beneath our feet and in the built environment all around us.
Our use of Earth’s minerals stretches beyond building with raw rock, expanding to use of the metals embedded within. The metals we rely so heavily upon can trace their origins to the nuclear fusion of exploding stars, the dust of which molded the Earth itself. Many of these metals (Cu, Zn, Pb, Ag, etc.) are accumulated around deep underwater vents, only brought to the surface and made accessible through tectonic collisions or when volcanoes erupt and form islands above the oceans, while others (Fe) were accumulated in the oceans before photosynthesizing organisms oxygenated the atmosphere. People living in areas with these metals available were given an advantage in developing successful civilizations through “revolutionized settlements, agriculture, and warfare” in much the same way that countries today rely on stores of precious and rare metals for devices we utilize daily. As Dartnell points out, humans moved from the Iron Age to Space Age in just 3,000 years!
The movement of these precious resources across continents and between empires was guided by the geography of the Earth beneath them. The lateral bands of desert and grassland created by patterns of global atmospheric moisture circulation provided the perfect landscape for transcontinental transport of goods and people in the form of Eurasia’s Silk Road. The wealth transported along this path developed some of the largest empires the world has ever seen, though Dartnell demonstrates that the same conditions that built empires also brought them down. While the Silk Road moved goods along the harsh desert to stationary agricultural empires, the grasslands to the north harbored nomadic tribes (Huns, Vandals, Mongols, etc.) on horseback, able to quickly invade, raid, and eventually defeat settled empires. Only when agrarian states modernized their armies, were they able to repel nomadic warriors from the grasslands and continue their advancement.
While global atmospheric circulation patterns helped develop the intercontinental trade of the Silk Road, the same patterns later brought trade to the global scale though facilitating sea travel. The latitudinal bands of wind encasing the Earth (such as the easterlies and westerlies trade winds in the Atlantic Ocean) and the associated circular patterns of surface currents (such as the combo of North Equatorial Current, Gulf Stream, North Atlantic Current and the Canary Current in the North Atlantic), provided the mode of transportation for early sailors to the Americas. Similar winds and currents powered by the seasonal monsoons aided mariners plying the trade routes to and from India and rest of maritime Asia. Learning and mastering these overarching winds and currents through years of trial and error, sailors from Europe and Asia gradually, mile by mile, traversed the Earth. The settlements established and resources moved along the way, set the stage for today’s globalization.
Winds and currents may have moved humans across oceans, but the stored energy beneath the Earth’s surface presented the final step in how the Earth made us who we are today. Dartnell describes the causes of the creation of coal, oil, and natural gas, which has defined human existence since the industrial revolution. The energy humanity relies so heavily upon is a product of a lack of decomposition of biomass and its long-term preservation under specific geological conditions. He concludes by detailing how, when viewed from space, the light we produce at night coincides with hotspots of past geological phenomena that have guided humans to where they now harness the energy from “fossilized sunshine”.
[1568317009].jpg)
Civilization’s Global Energy Use Footprint: Composite global view of Earth at night compiled from over 400 satellites. Brightest areas are the most urbanized (representing intensity of energy use), but not necessarily the most populated (compare western Europe and Eastern U.S. with China and India). Image credit: NASA/NOAA 2017.
So, what brought you to this exact moment? Dartnell shows through the pages of Origins: How the Earth Made Us how a series of fortunate Earthly events have gotten us to this moment. For the reader who is curious about the world around us, and about what serendipitous events brought us to where we are today, this book is sure to satisfy. “The Earth set the stage for the human story, and its landscapes and resources continue to direct human civilization,” Darnell writes. Just as it has done for billions of years, the Earth will no doubt continue to shape our future in ways yet unknown to us. Surely, there is much more to the answer for the question of “How did we get here?” than the standard text books will provide. Planetary sciences often take a backseat in public education. Dartnell’s tome shows that knowledge such as what lies beneath our feet and what is happening around us is vital – especially in a period of polarized geopolitics and a climate system that is changing faster than ever before in the history of human civilization. As “children of plate tectonics” who find ourselves in this “inter-glacial interim civilization”, one thing is certain: the changing Earth continues to change us.
“The future is already here – its just unevenly distributed”
– Michael Gibson, 2019.
Literature Cited:
- Gibson, Michael. 2019. In Futurology: Navigating the Rapids. The Economist, July 6th, 2019.
- Milankovitch, M. 1941. Canon of Insolation and the Ice-Age problem.
- Talking Heads, 1980. In Once in a Lifetime, Sire Records, Warner Brothers Records Inc.

